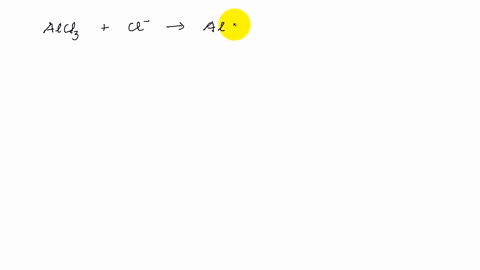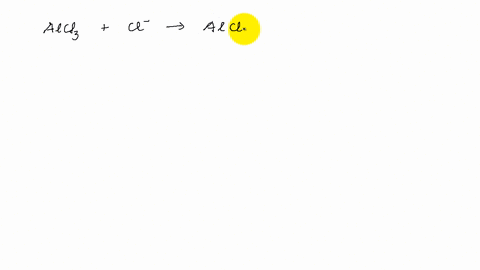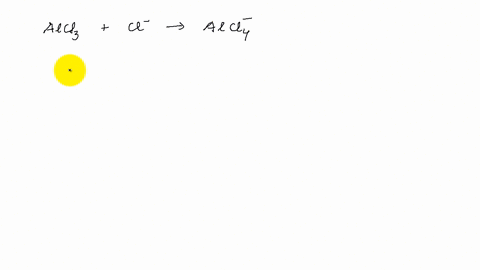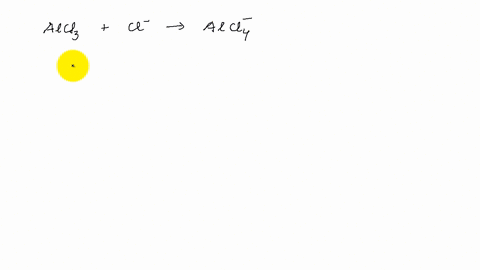AlCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, bond angle | Molecular geometry, Molecular, Molecular shapes

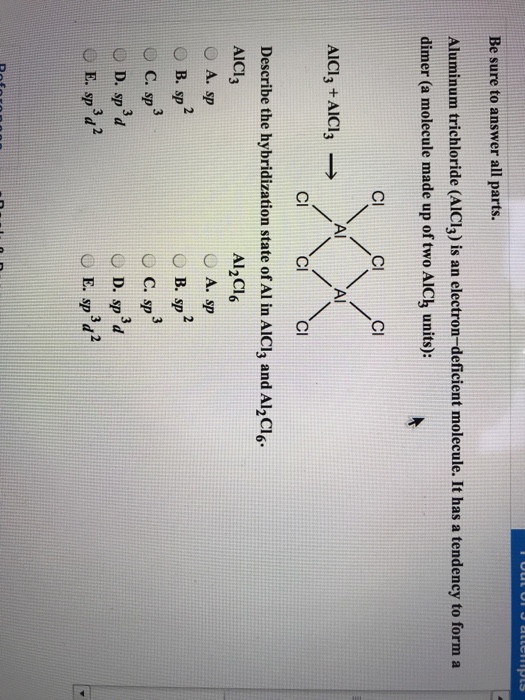
What is the molecular structure of the molecule AlCl3? What characteristic is notable about the structure that helps us to understand the acidic nature of AlCl3? Write a chemical reaction between AlCl3
When BCl3 is treated with water, it hydrolyses and forms [B[OH]4]^– only whereas AlCl3 in acidified aqueous solution forms [Al (H2O)6]^3+ ion. - Sarthaks eConnect | Largest Online Education Community

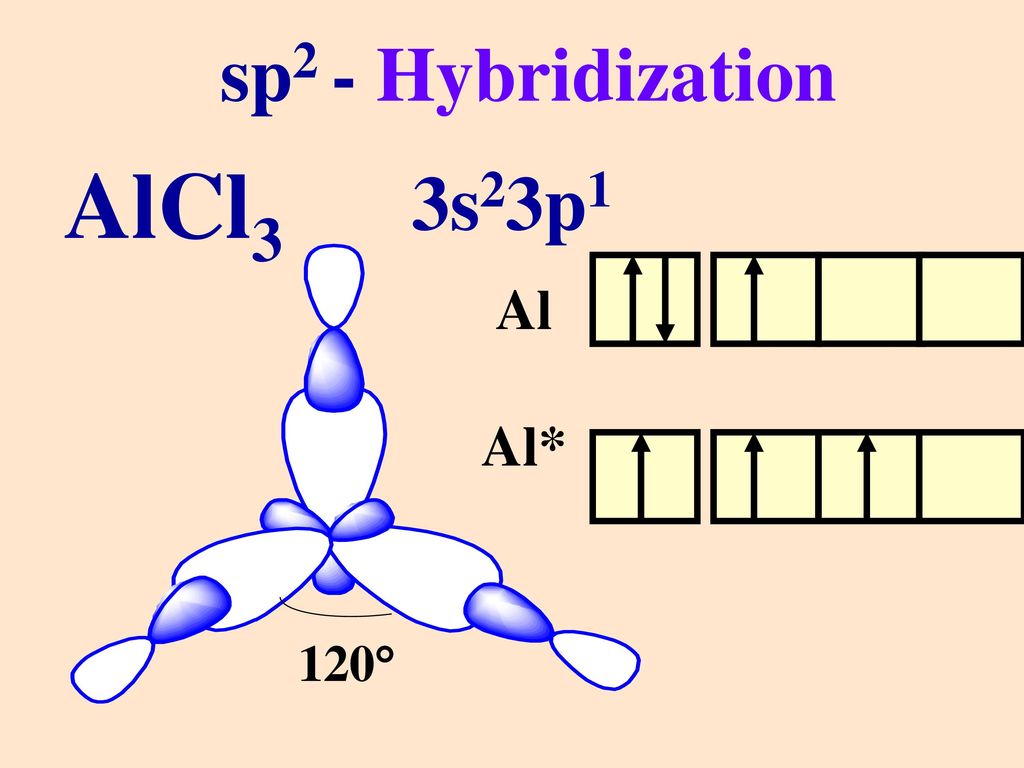
AlCl3 Lewis Structure, Molecular Structure, Hybridization, Bond Angle, and Shape - Geometry of Molecules

The change in hybridization of aluminium when `Al_(2)Cl_(6)` decomposes in the gas phase is : - YouTube

AlCl3 Lewis Structure, Molecular Structure, Hybridization, Bond Angle, and Shape - Geometry of Molecules

SOLVED: Which molecule has a central atom that is sp3 hybridized? Select the correct answer below: SF6 CH3Cl AlCl3 PCl5
6. Which one of the following compounds has the electron pair geometry as the trigonal bipyramid with three equatorial positions occupied by lone pairs of electrons? A) [AlCl3] B) XeF2 C) [
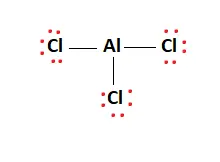
AlCl3 Lewis Structure, Molecular Structure, Hybridization, Bond Angle, and Shape - Geometry of Molecules

Describe the change in hybridization of the Al atom in the following reaction Alcl3+Cl- gives AlCl4- - YouTube