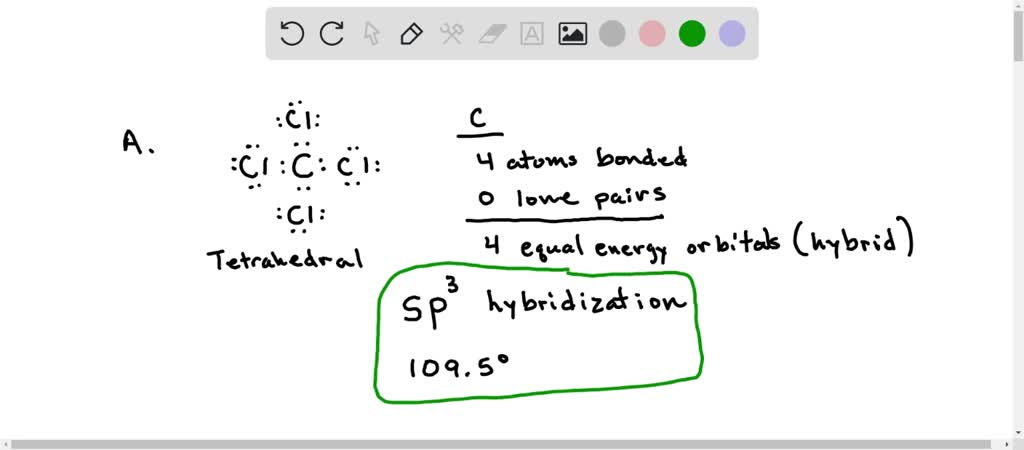
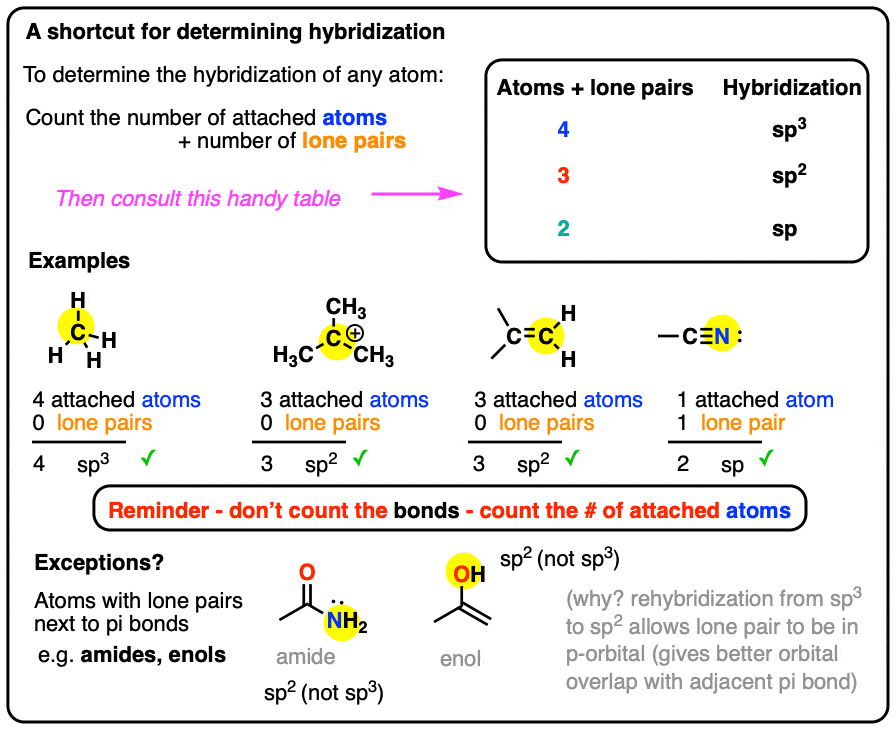
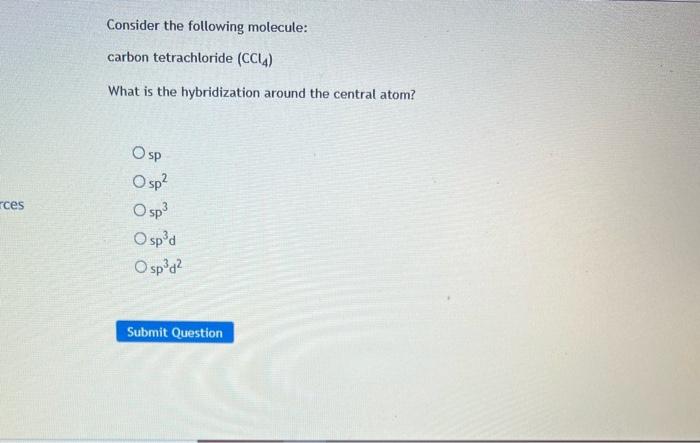
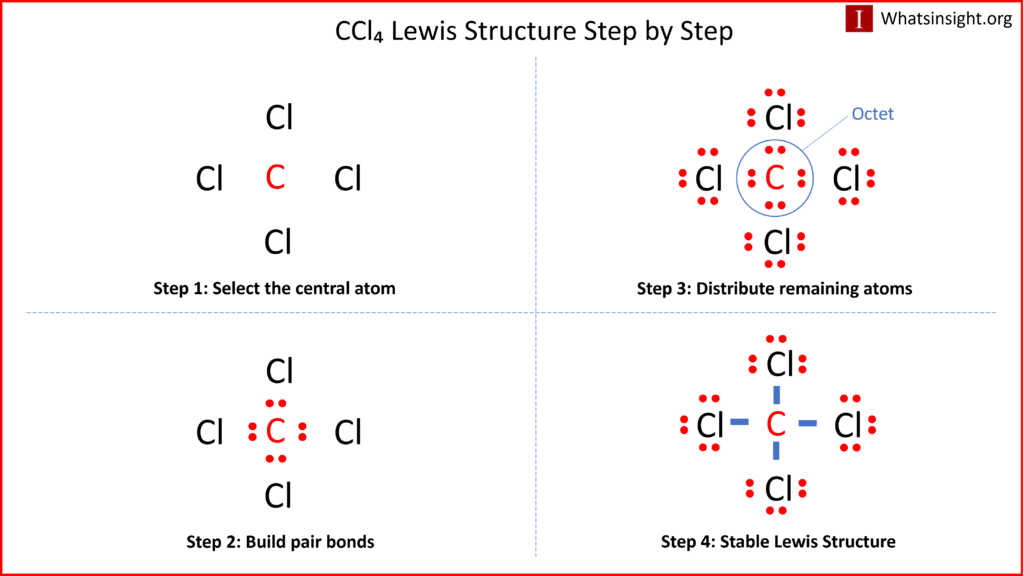
SOLVED: The hybridization of the carbon atom in the carbon tetrachloride molecule (CCl4) is of the type sp3.

Molecular geometry of CCl4 | Hybridisation of CCl4 | Carbon tetrachloride structure and geometry - YouTube

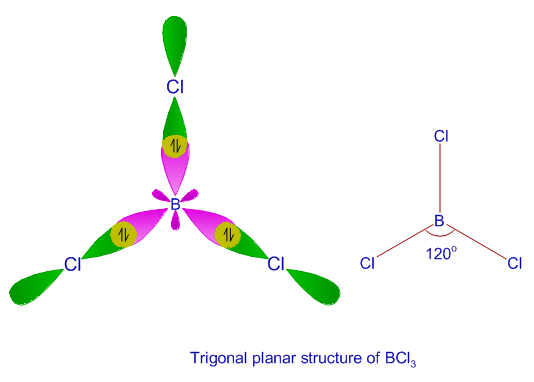
State hybridization in Bcl3 molecule - Chemistry - Chemical Bonding and Molecular Structure - 12421859 | Meritnation.com
Chemokine-mediated inflammation in the degenerating retina is coordinated by Müller cells, activated microglia, and retinal pigment epithelium | Journal of Neuroinflammation | Full Text

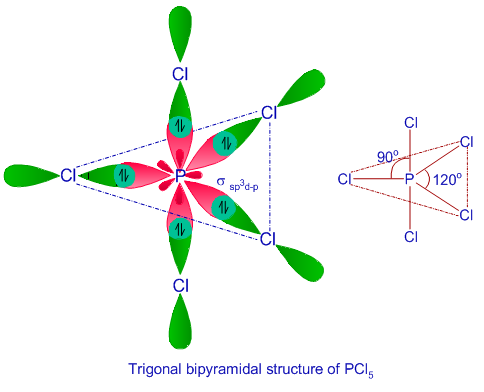
Starting from Lewis structure, determine the hybridization types of the central atom of TeCl4 and ICl4 .