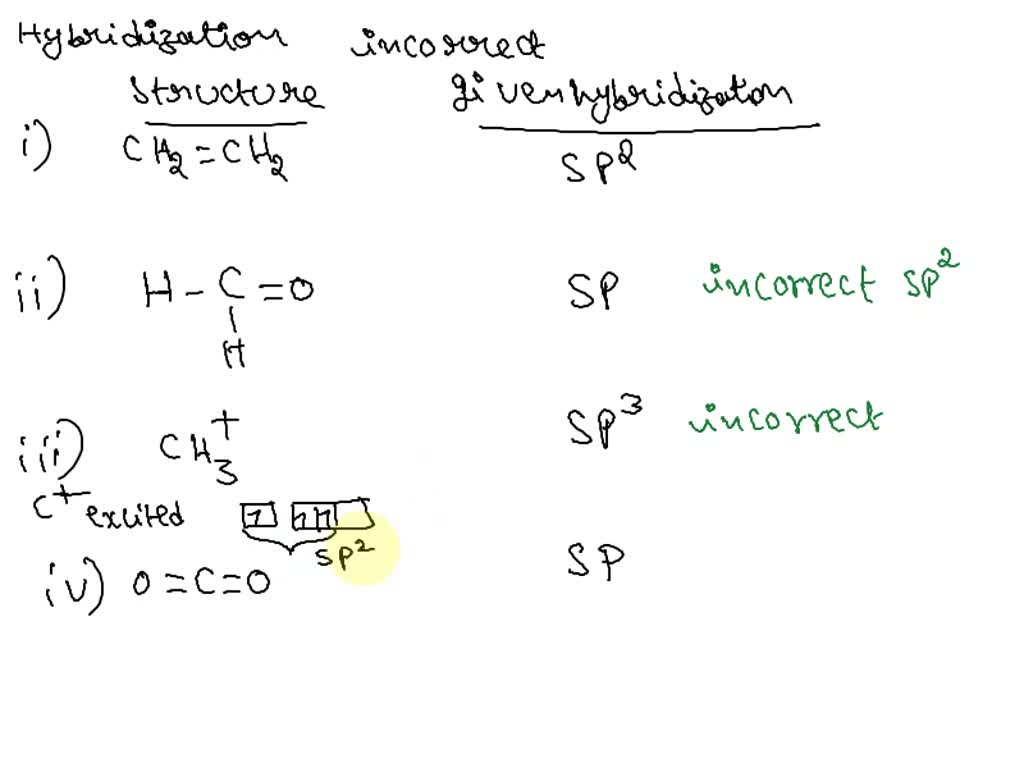
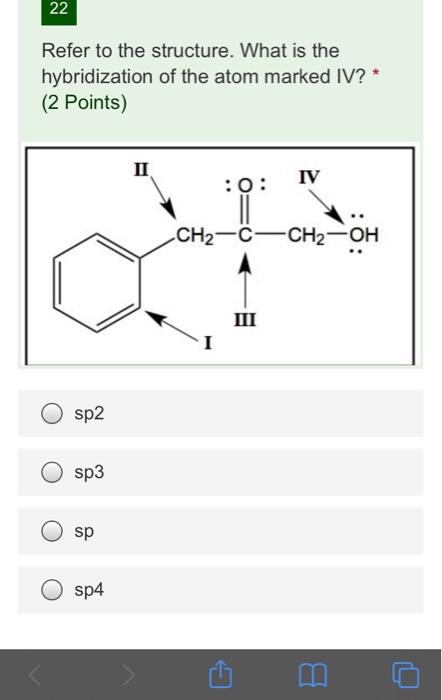
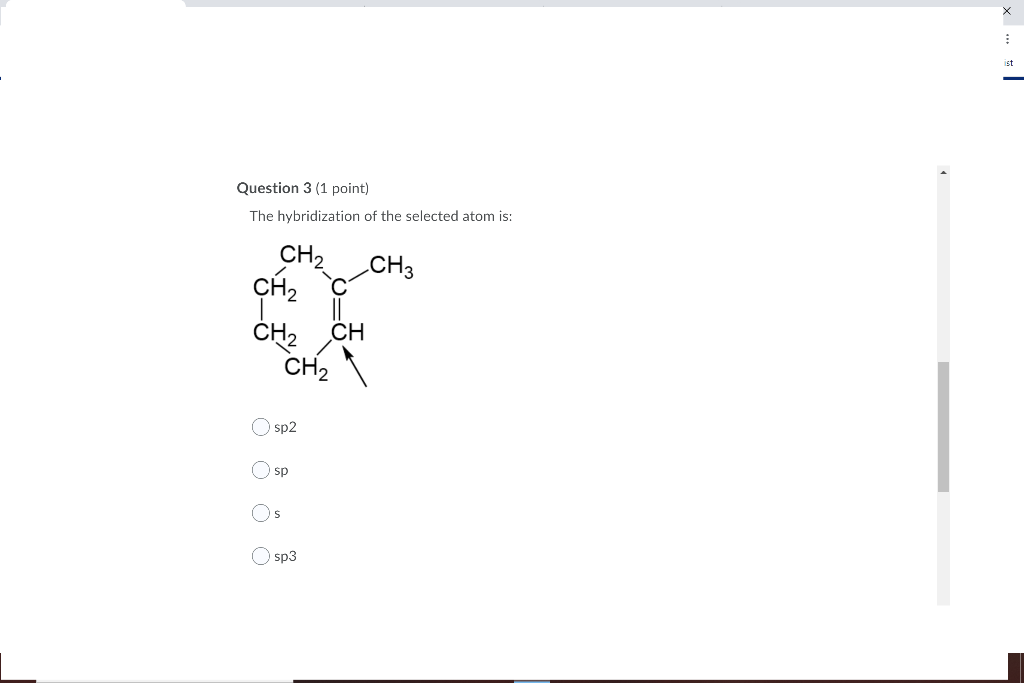
Write the type of hybridisation of each of the carbon atom in the following structures: (i) ch2=c=ch2 (ii) ch3 ch=ch ch3

What are hybridisation states of each carbon atom in the following compounds ?CH2=C=O, CH3CH=CH2, - YouTube

SOLVED: Specify the hybridization of the carbon atom in C=CH2. In the molecule XeF2, how many pairs of electrons surround Xe and what is the molecular geometry? What is the energy of
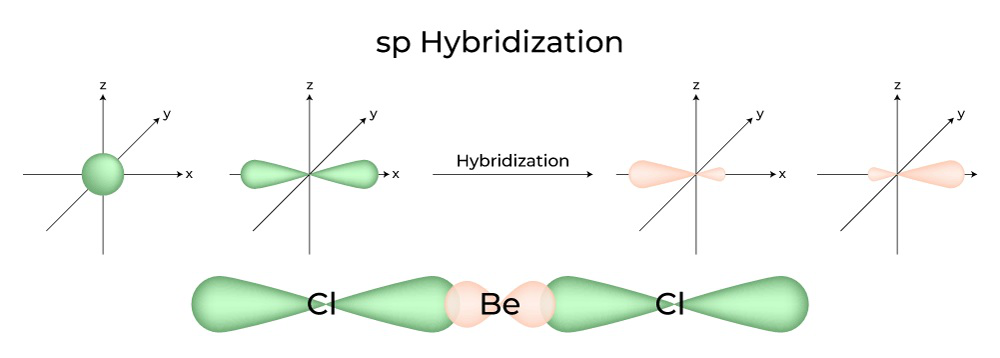
/chapter2/pages1and2/page1and2_files/sp2hyb.png)





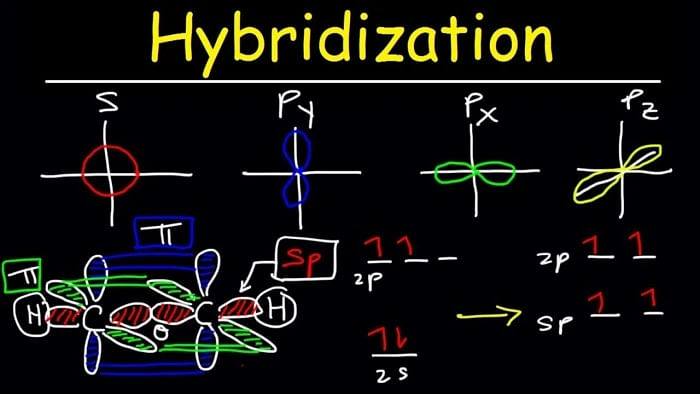





.jpg?revision=1&size=bestfit&width=601&height=192)