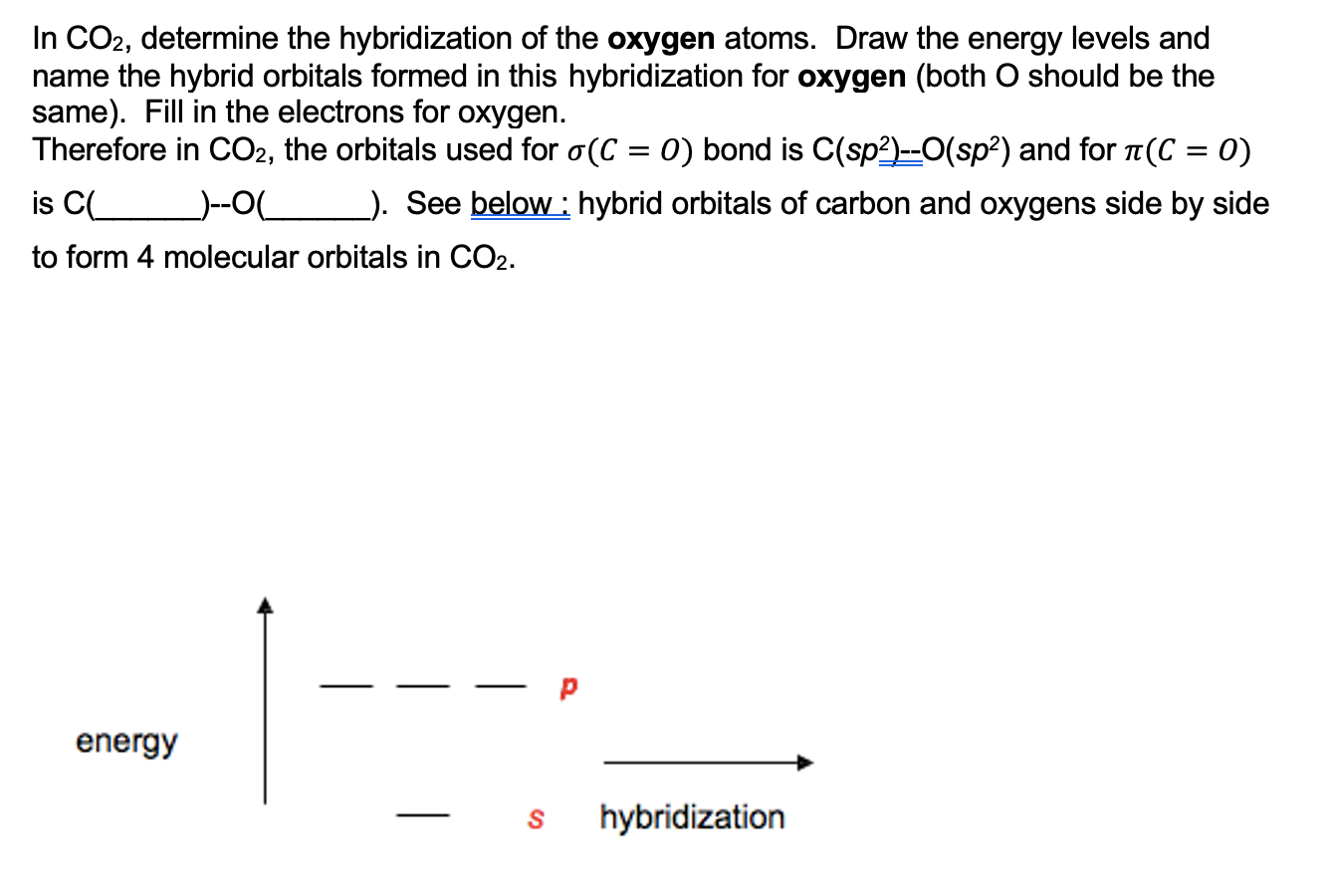
SOLVED: Which one of the following statements about orbital hybridization is incorrect? M a. The carbon atom in CH4 is sp3 hybridized. b. The carbon atom in CO2 is sp hybridized. c.
✓ Solved: Describe the bonding on the carbon atom in carbon dioxide, CO2, using valence bond theory.
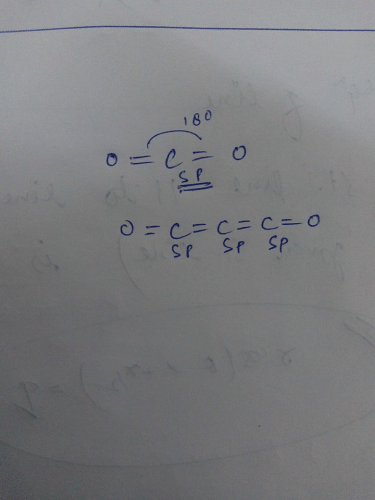
Hybridization of carbon C3O2 and CO2 are:a)sp in bothb)sp and sp2c)sp2 and spd)sp3 and spCorrect answer is option 'A'. Can you explain this answer? - EduRev Chemistry Question

The molecule CO2 has two C-O double bonds. Describe the bonding in the CO2 molecule. Which involves hybrid - brainly.com

Draw a Lewis structure and an orbital picture for carbon dioxide, CO2. What kind of hybridization does the carbon atom have? What is the relationship between CO2 and allene? | Homework.Study.com

Allene is structurally related to carbon dioxide, CO2. Draw a picture showing the orbitals involved in the sigma and pi bonds of CO2, and identify the likely hybridization of carbon. | Homework.Study.com

p–d Orbital Hybridization Induced by p-Block Metal-Doped Cu Promotes the Formation of C2+ Products in Ampere-Level CO2 Electroreduction | Journal of the American Chemical Society
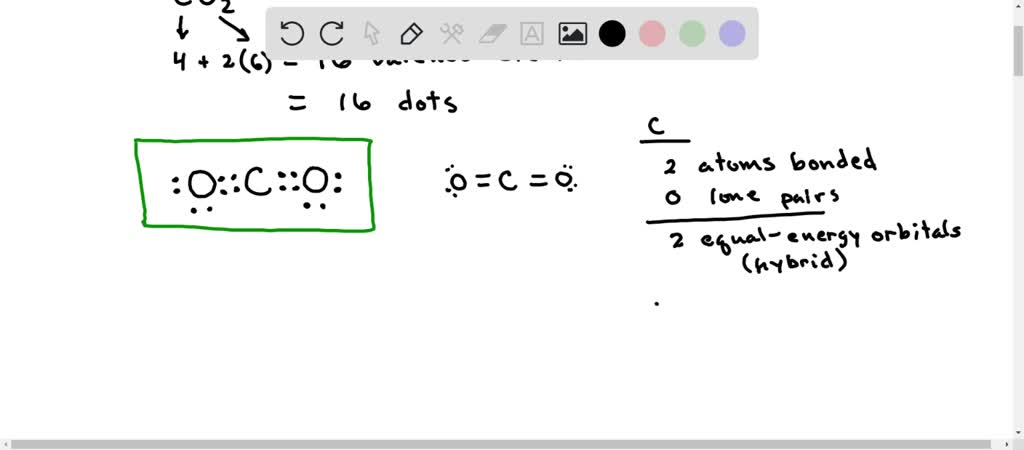

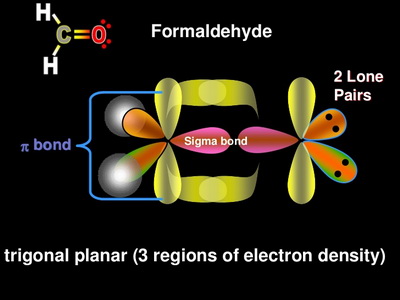


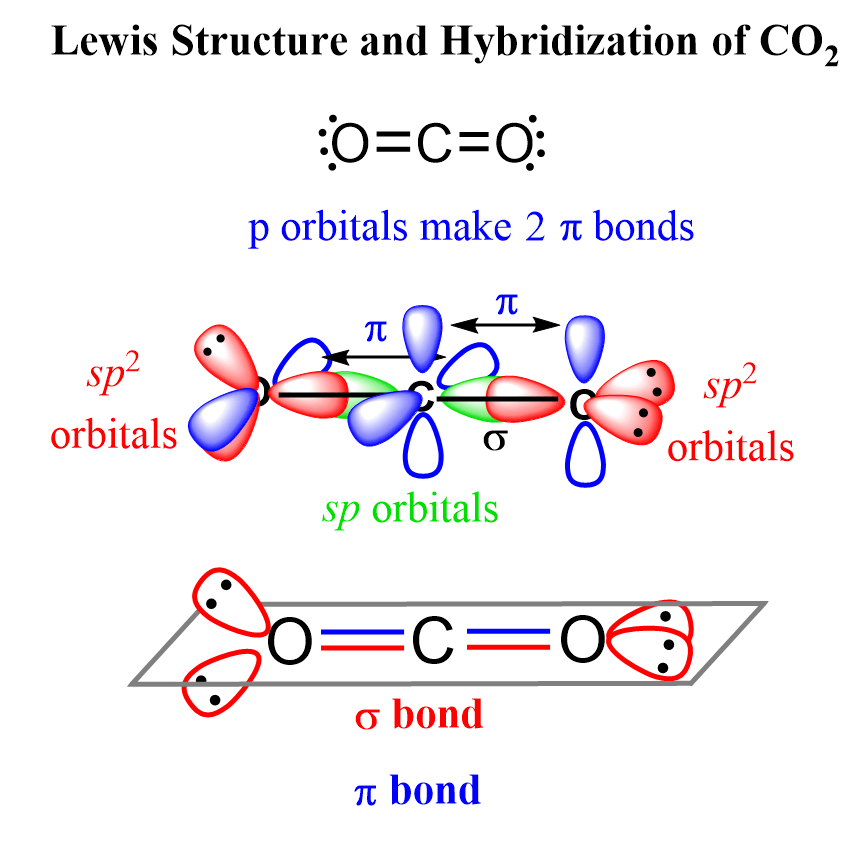

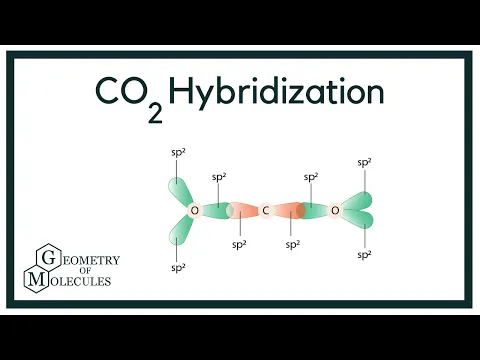


![SOLVED] In CO2 hybridization of carbon is sp sp2 sp3 None of t - Self Study 365 SOLVED] In CO2 hybridization of carbon is sp sp2 sp3 None of t - Self Study 365](https://static.tllms.com/moodle-migration/47309_7fc775895c5b6da80cbe1e11e0d869d746d07138_S-1.PNG)