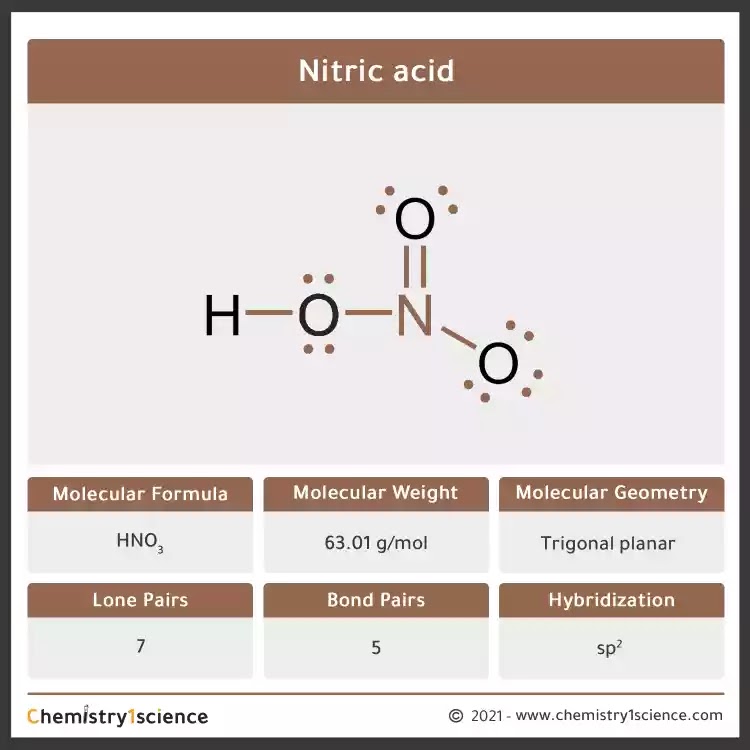
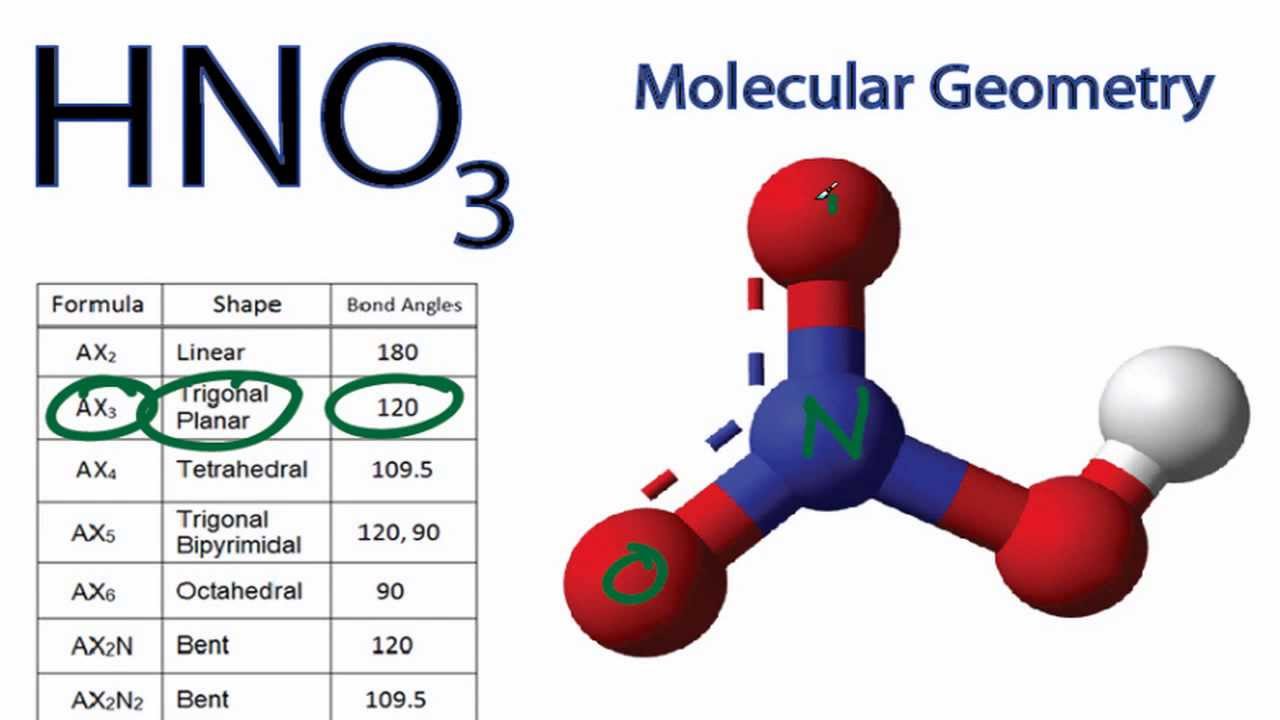
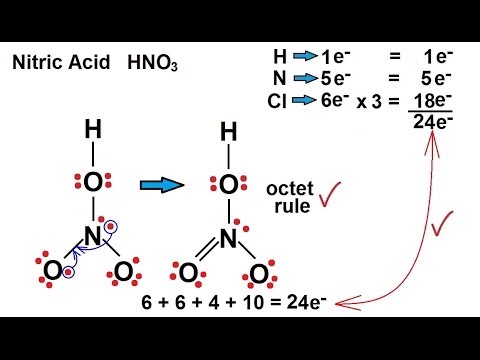
Provide the following information for the compound HNO3. a. Lewis dot structure b. hybridization c. electron geometry d. molecular geometry e. polarity | Homework.Study.com

SOLVED: What is the electron-pair geometry about the nitrogen atom in the HNO3 (nitric acid) molecule. Hint: The nitrogen atom is the central atom and the hydrogen atom is attached to an
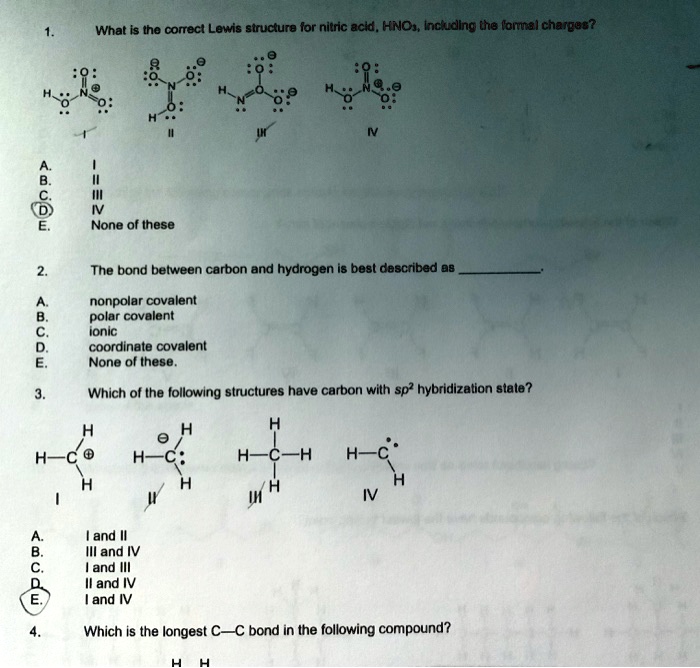
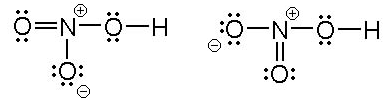
SOLVED: What is the correct Lewis structure for nitric acid, HNO3, including the formal charges? None of these. The bond between carbon and hydrogen is best described as nonpolar covalent. Which of

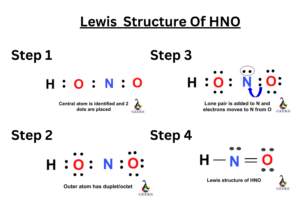
Nitric acid (HNO3) Lewis structure, molecular geometry, hybridization, polar or nonpolar | Molecular geometry, Molecular, Electron configuration