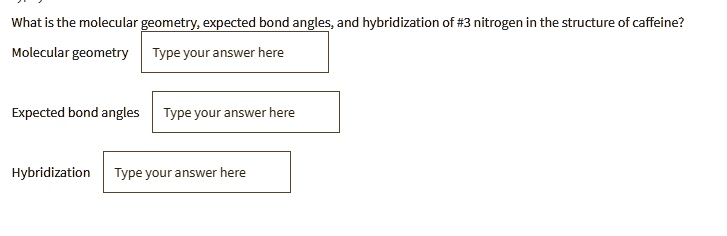
SOLVED: What is the molecular geometry, expected bond angles, and hybridization of nitrogen in the structure of caffeine? Molecular geometry: Type your answer here Expected bond angles: Type your answer here Hybridization:
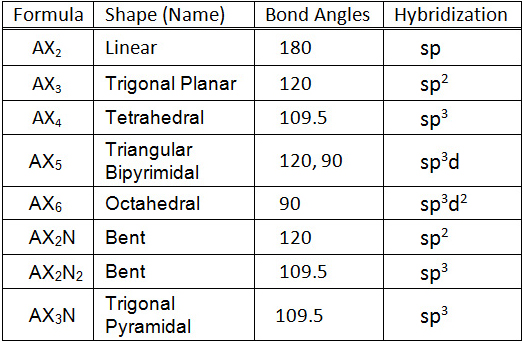
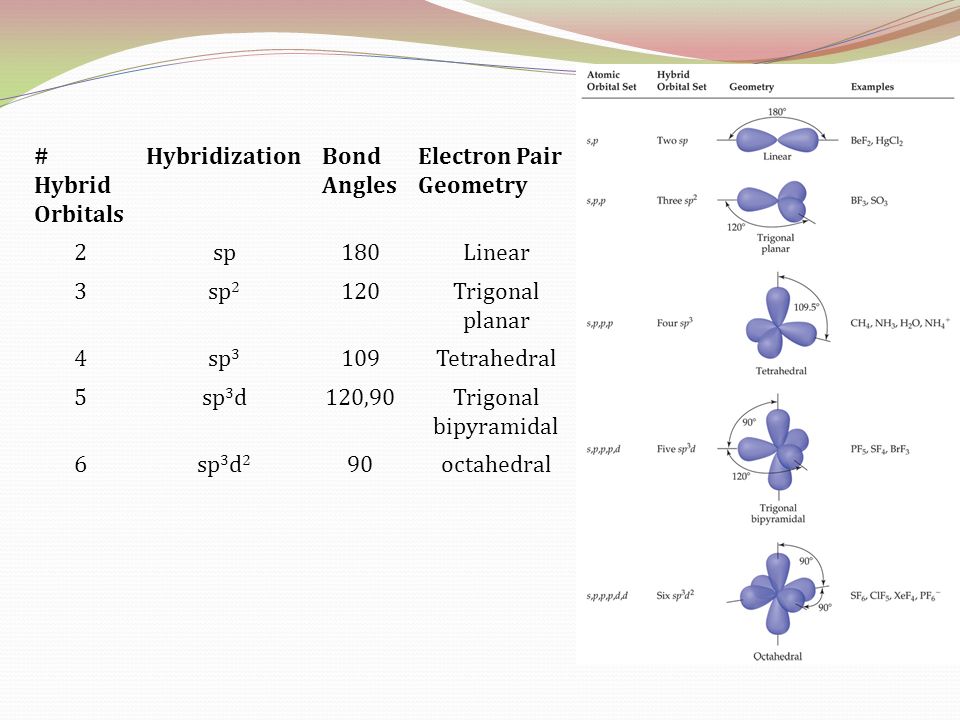
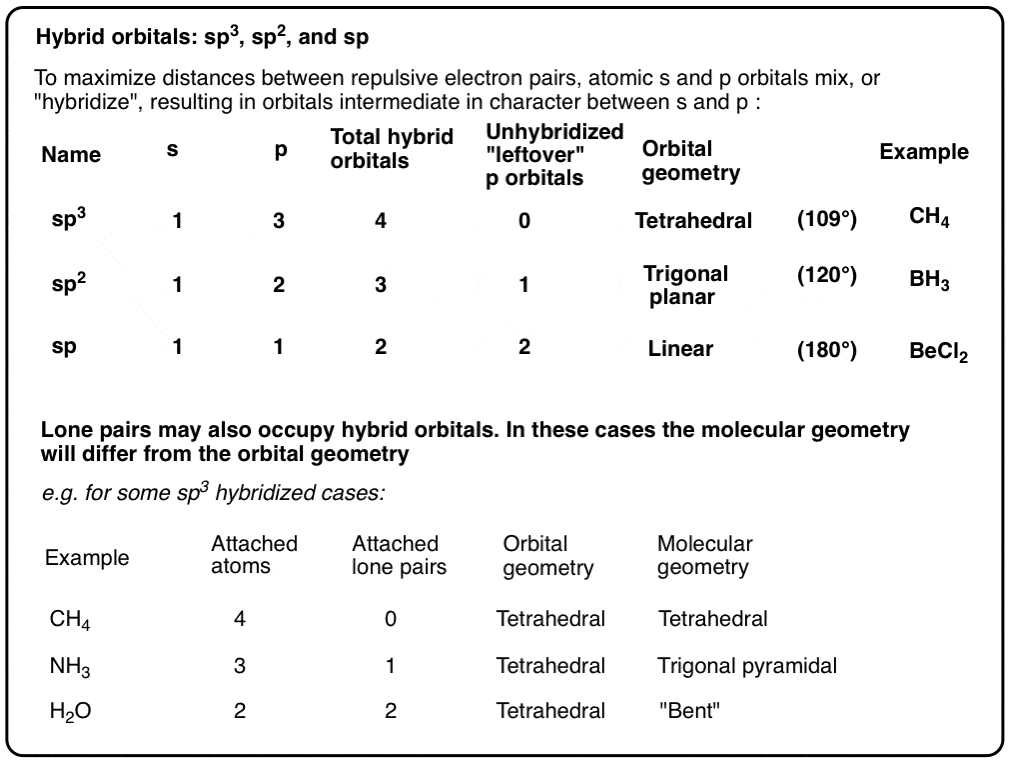
What mode of hybridization is associated with each of the five common electron domain geometries? | Socratic

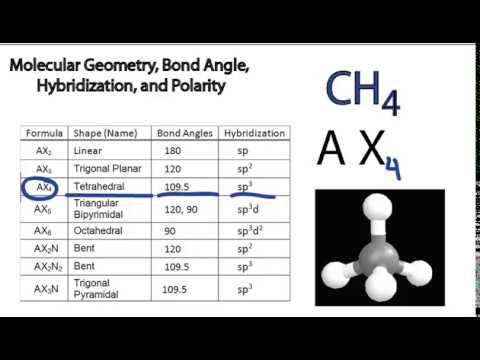
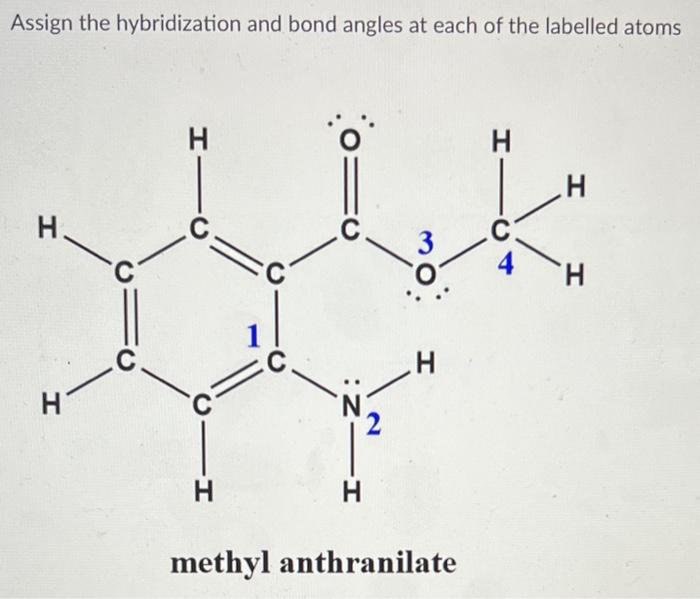
For each of the following compounds: a) Give the hybridization and approximate bond angles around each atom except hydrogen. b) Draw a three-dimensional diagram, including any lone pairs of electrons. | Homework.Study.com
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora

Table 2. Hybridization, Molecular Geometry and Bond Angles without/with lone pair of electrons : Innovative Mnemonics Make Chemical Education Time Economic – A Pedagogical Review Article : Science and Education Publishing

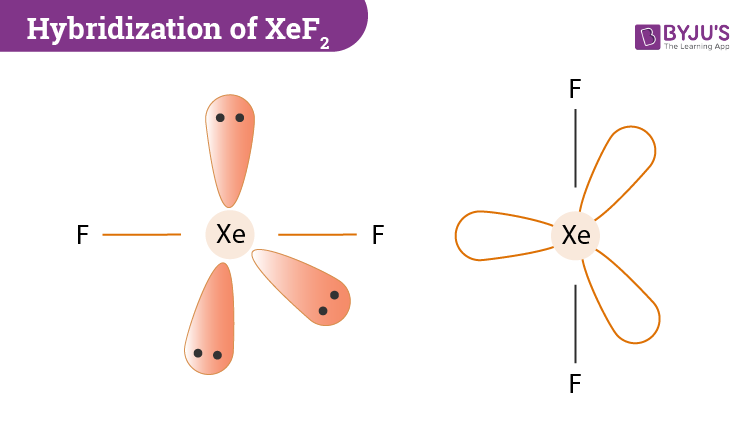
SOLVED: What is the orbital hybridization and bond angles for the set of hybrid orbitals represented below? sp3d2, 90°, 120°, 180° sp2d2, 90°, 180° sp3d, 90°, 120°, 180° sp3d2, 90°, 180° sp3d,

For each of the following compounds: a) Give the hybridization and approximate bond angles around each atom except hydrogen. b) Draw a three-dimensional diagram, including any lone pairs of electrons. | Homework.Study.com

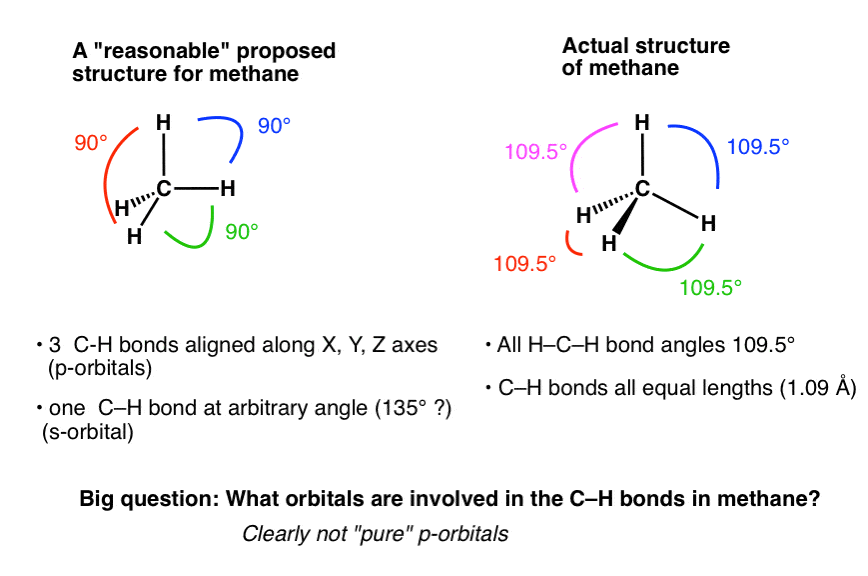

![ANSWERED] A What is the hybridization of the central atom in BeCl - Kunduz ANSWERED] A What is the hybridization of the central atom in BeCl - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20220509230100949162-4415097.jpg)