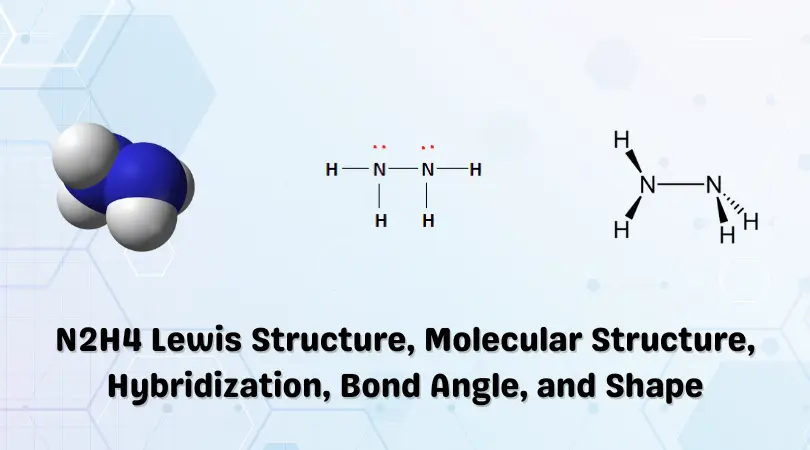
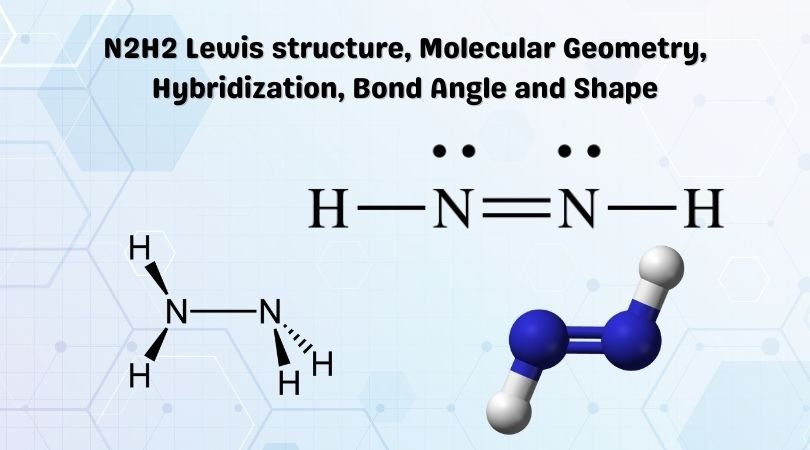
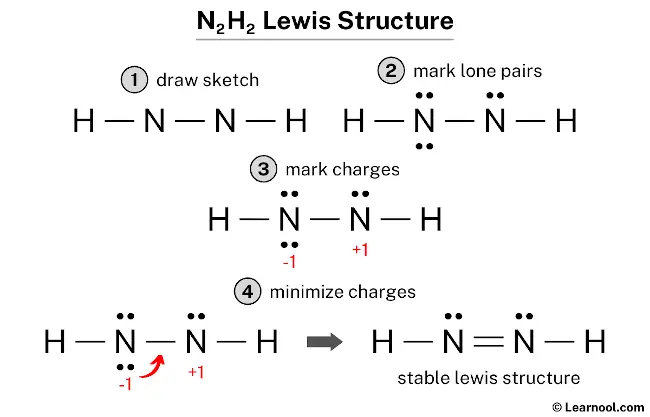
N2H2 Molecular Geometry, Bond Angles & Electron Geometry (Diimide) | Molecular geometry, Molecular, Geometry

SOLVED: Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping orbitals, and label

SOLVED: What is the hybridization of each nitrogen in the N2H2 molecule? The Lewis structure is given. A-N=N-H
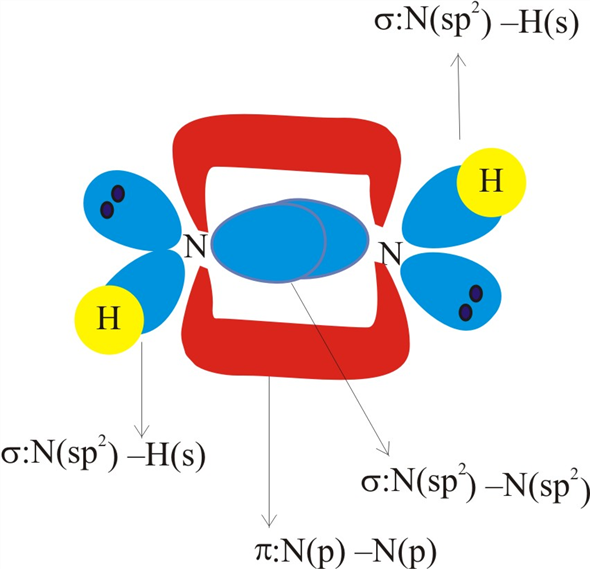
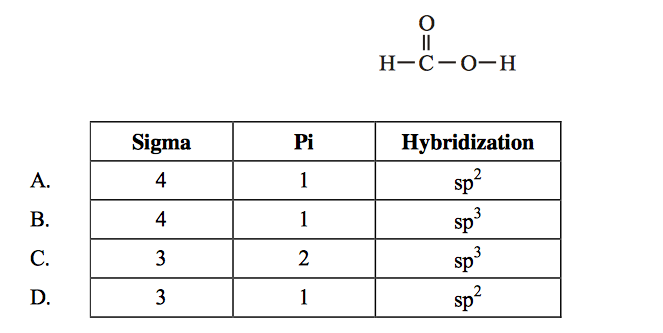
Solved: Chapter 10 Problem 35E Solution | Masteringchemistry -- Standalone Access Card -- For Principles Of Chemistry 2nd Edition | Chegg.com

![14.2/S2.2 .16 Lewis, hybridization (sp3,sp2,sp) , shapes and angles [HL IB Chemistry] - YouTube 14.2/S2.2 .16 Lewis, hybridization (sp3,sp2,sp) , shapes and angles [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/x1fUDQKztsY/maxresdefault.jpg)