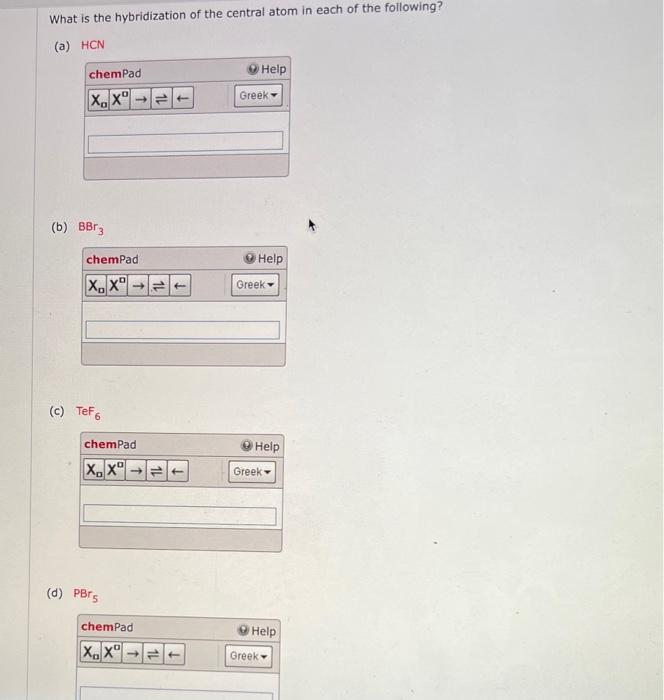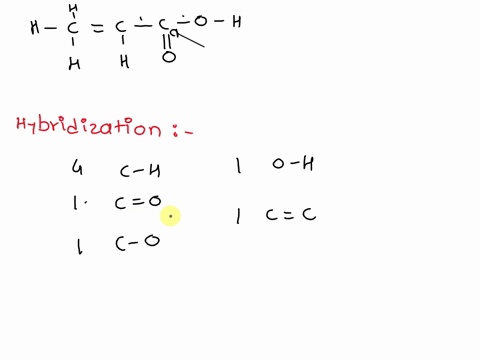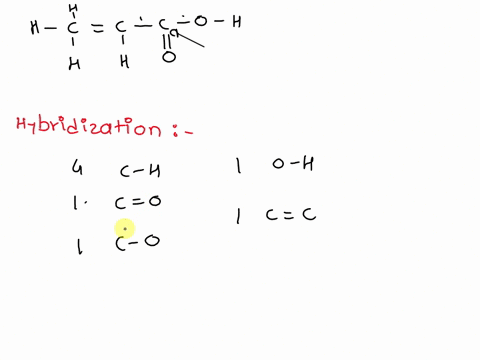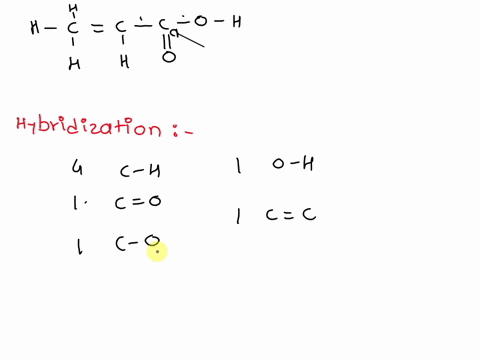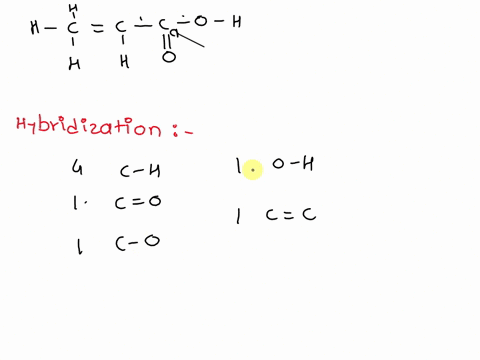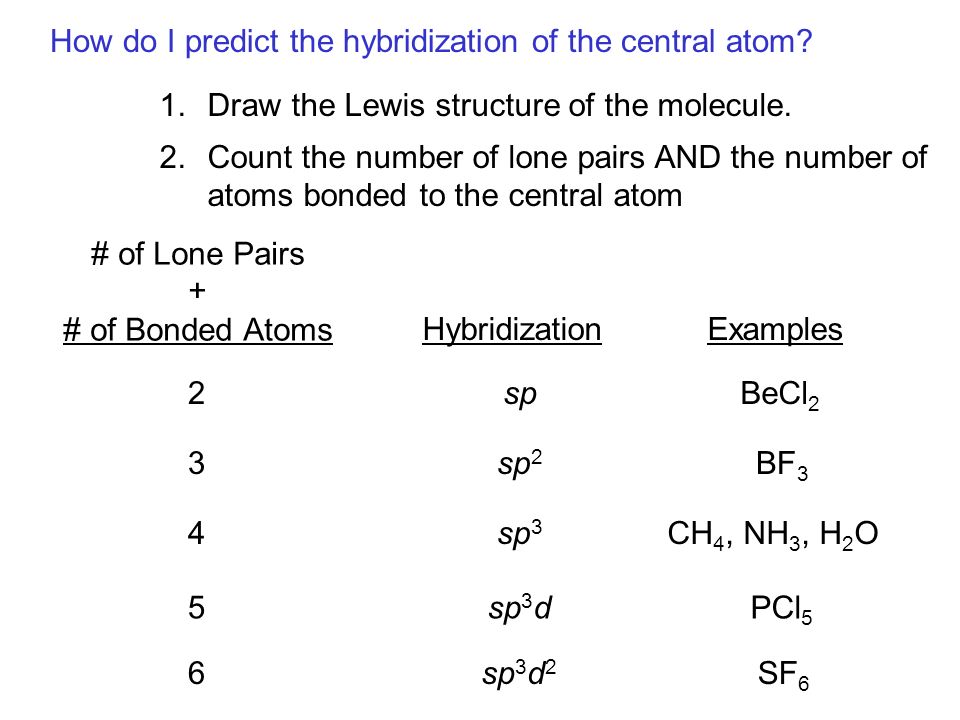
Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals Chapter ppt video online download

Types of Hybridization: Definitions, Examples, Key Features, Steps to Determine, Shapes, and Rules | CollegeSearch

SOLVED: PCl5 and PBr5 exist in sp^3d hybrid state in gaseous phase but in solid state, which of the following statement is true? A. P in PCl5 exists in sp^3 - hybridization

12. Which of the following compounds exhibit d 2sp 3 hybridization? (Select all that apply.) BrF5 ClF5 KrCl4 XeCl2 PCl5 13. Draw the Lewis structure for IF3 and answer the following question.

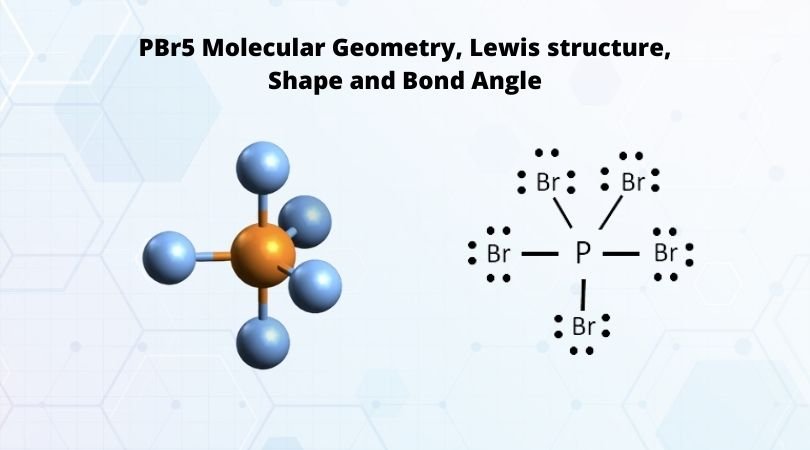
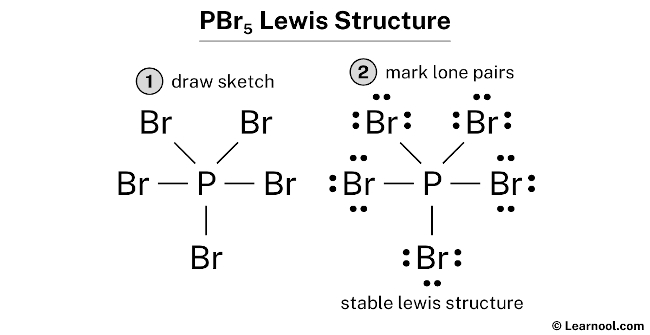
1. Explain the hybridisation and geometry of the following molecules. a. PBr5 b. ClF3 c. SF4 d. PH3 e. - Brainly.in

12. Which of the following compounds exhibit d 2sp 3 hybridization? (Select all that apply.) BrF5 ClF5 KrCl4 XeCl2 PCl5 13. Draw the Lewis structure for IF3 and answer the following question.
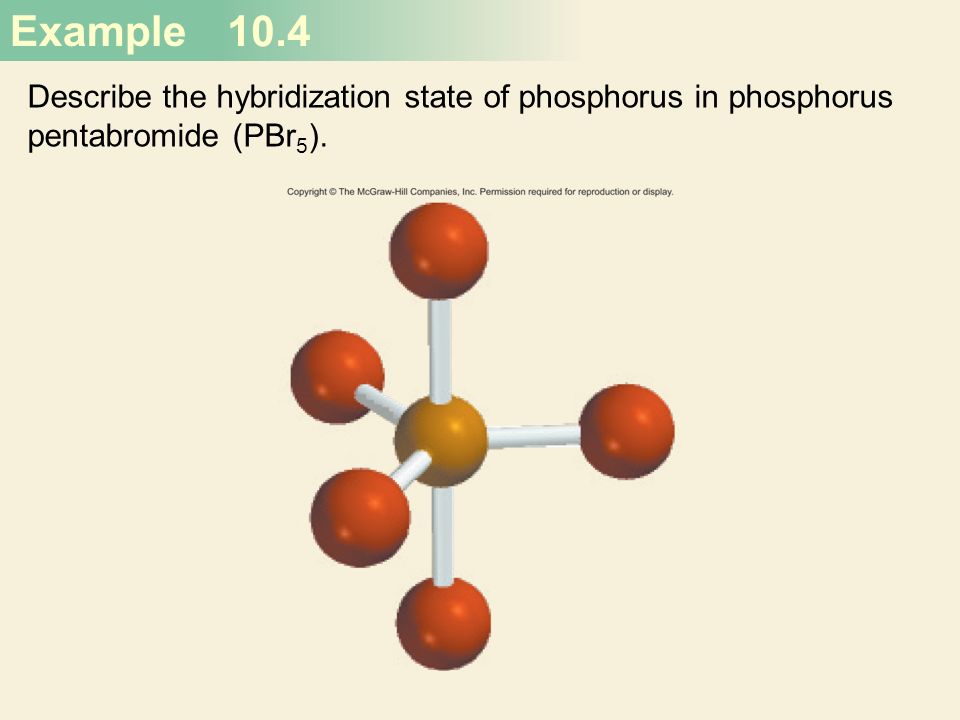
Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals Chapter ppt video online download