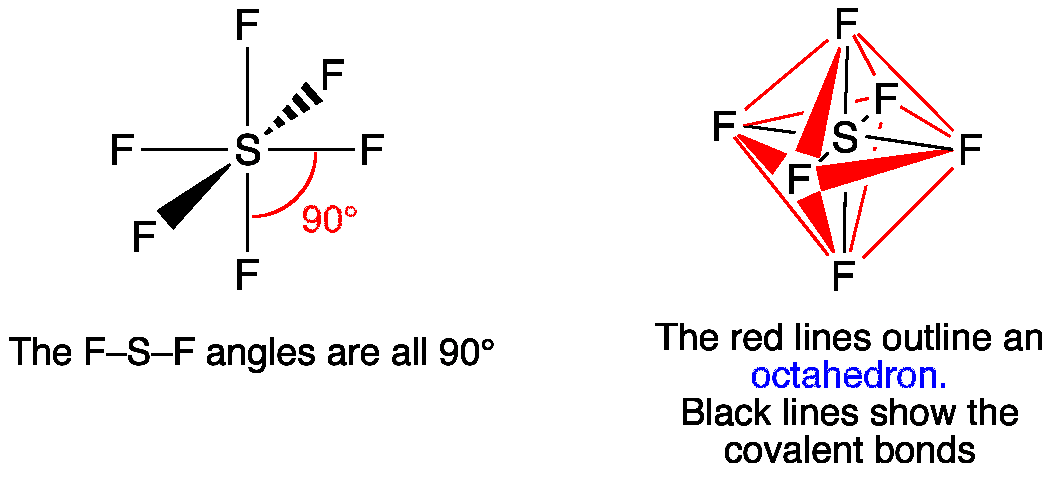
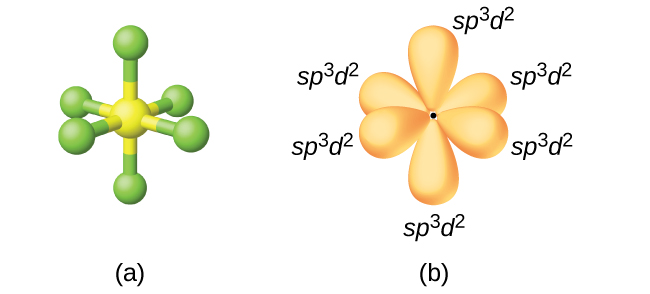
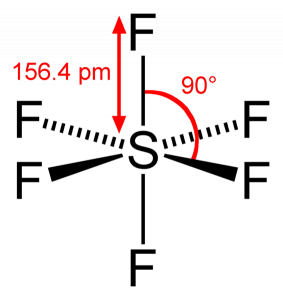
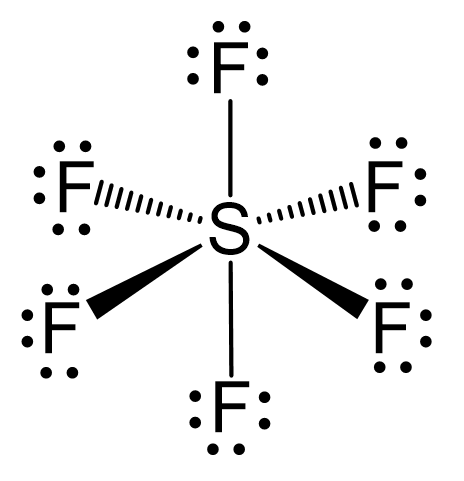
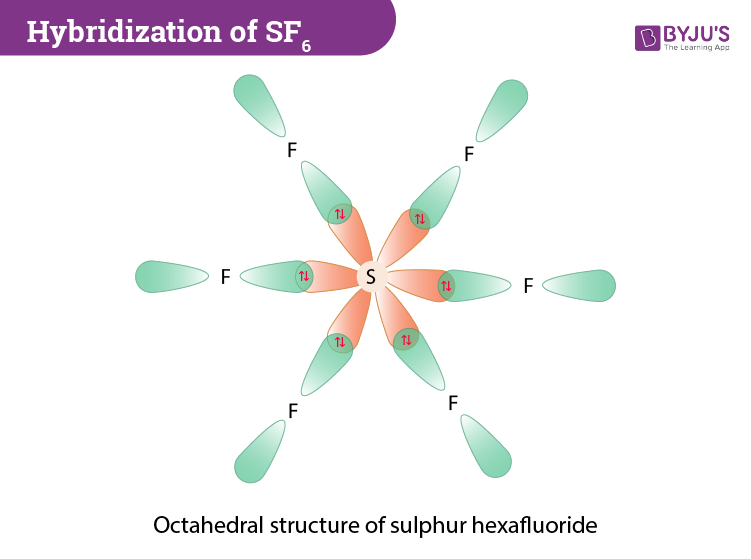
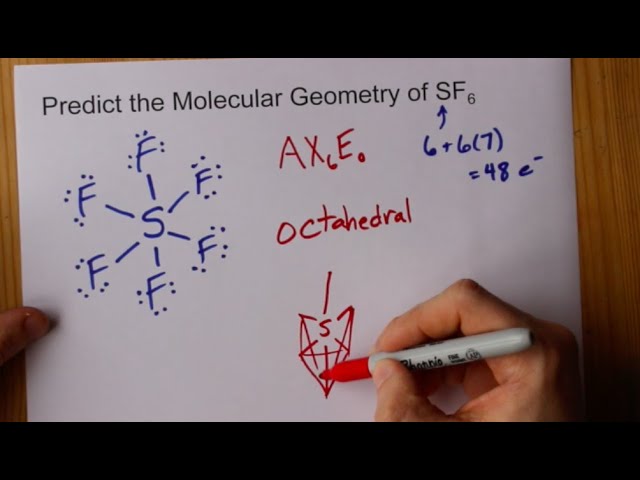
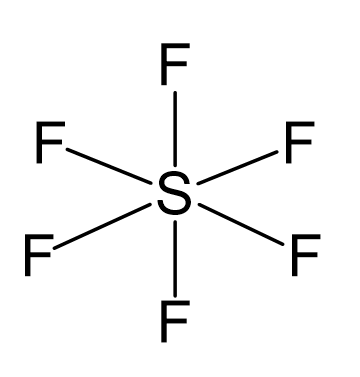
Atomic orbitals involved in hybridization of SF_{6} molecule:3s,3p_{x},3p_{y},3p_{z},3d_{{z}^{2}},3d_{{x}^{2}-{y}^{2}}3s,3p_{x},3p_{y},3p_{z},3d_{{z}^{2}}4s,4p_{x},4p_{y},4p_{z},3d_{{x}^{2}-{y}^{2}},d_{xy}3s,3p_{x},3p_{y},3d_{xy},3d_{yz},3d_{xz}

Chemistry - Molecular Structure (35 of 45) s-p3-d2 Hybridization - Sulfur Hexafloride - SF6 - YouTube

The sp3d2 Hybridization and Octahedral Geometry | Molecular geometry, Chemistry, Electron configuration

In which of the following the central atom is sp3 hybridised?a)NH4+ b)BF3c) SF6 d)PCl5Correct answer is option 'A'. Can you explain this answer? - EduRev NEET Question
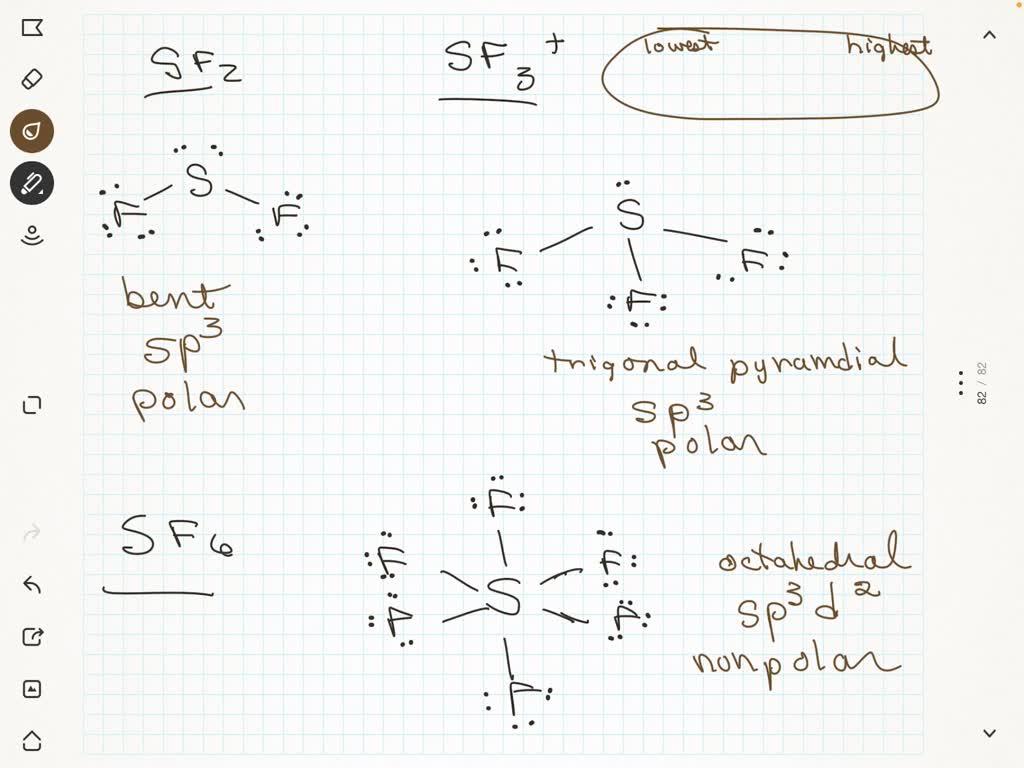
SOLVED: Consider the following molecules: SF2, SF3+, SF6. Draw the Lewis dot structures, determine VSEPR shapes, give hybridization of the central atoms, and predict whether the molecules are polar. Put in order

For the molecule SF6, determine its (a) electronic geometry, (b) number of nonbonding domains on the central atom, and (c) polarity. | Homework.Study.com

Describe hybridisation in the case of PCl5 and SF6. The axial bonds are longer as compared to equatorial bonds in PCl5 whereas in SF6 axial bonds and equatorial bonds have the same
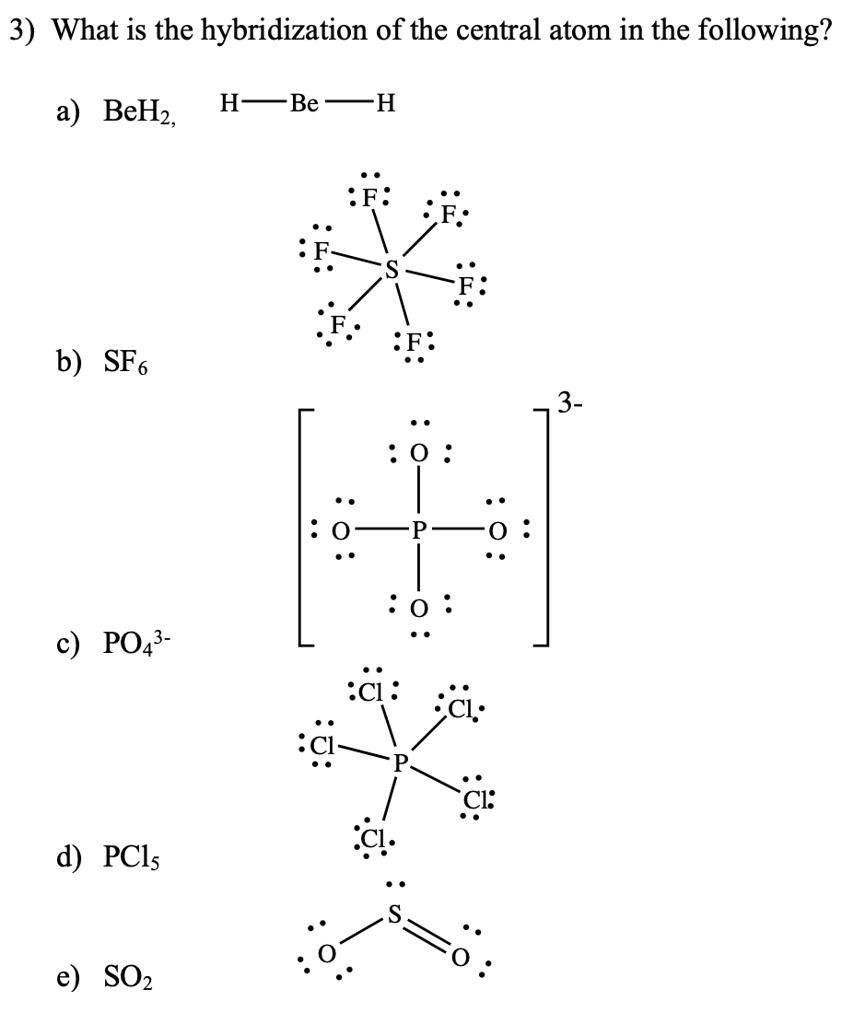
SOLVED: What is the hybridization of the central atom in the following? H2O a) BeH2, b) SF6 c) PO3^- d) PCl5 e) SO2