Total number of molecules which hydrolysed at room temperature and hybridization of central atom ... - YouTube
Hybridization is a phenomenon that takes place in an atom before chemical bonding. How is hybridization responsible for the observed structure of SiCl4? - Quora
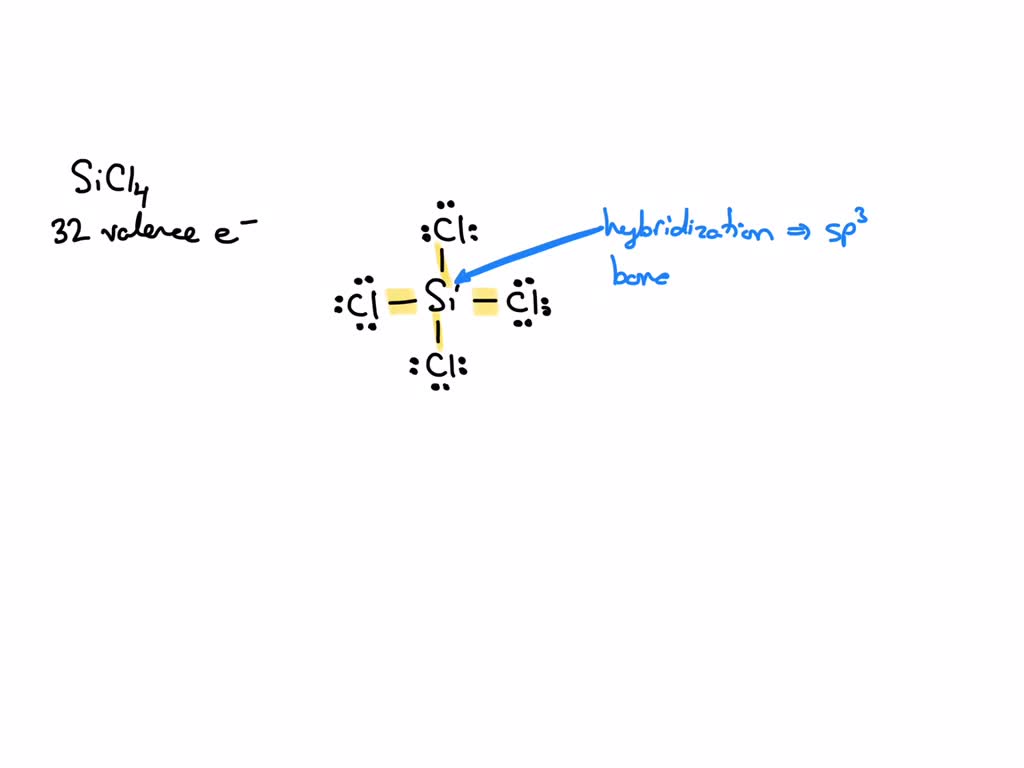
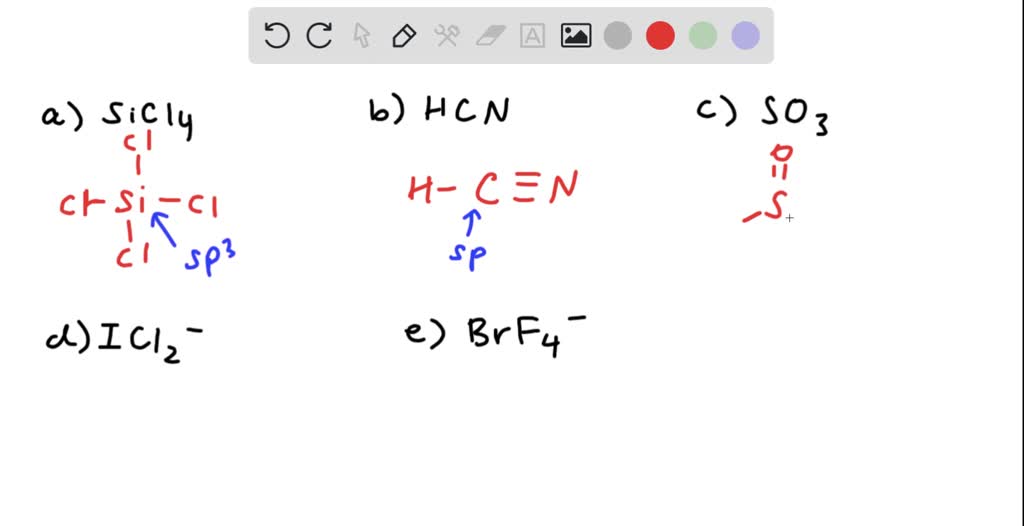
SOLVED: What is the hybridization of the central atom in SiCl4? Hybridization = What are the approximate bond angles in this substance? Bond angles = fill in the blank 2° B. What

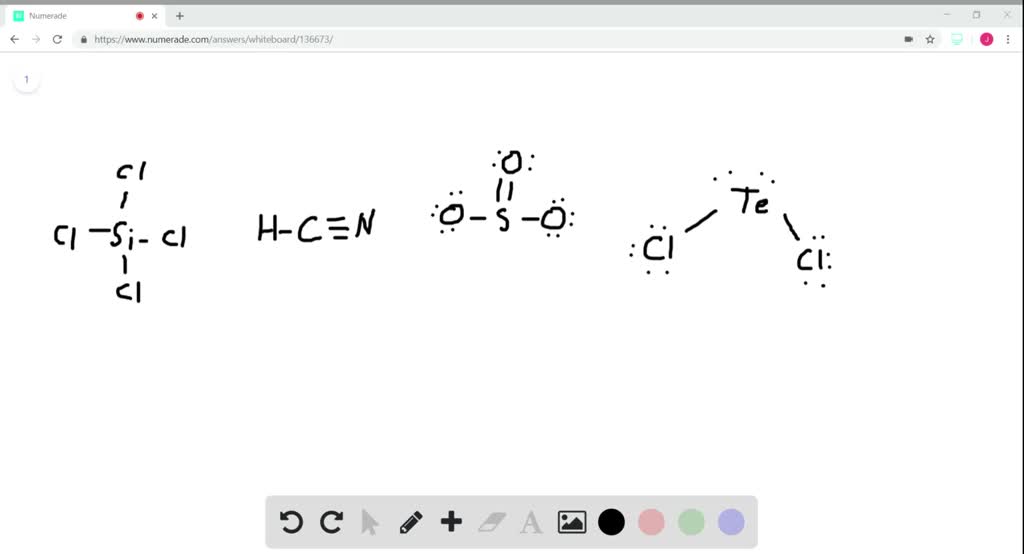
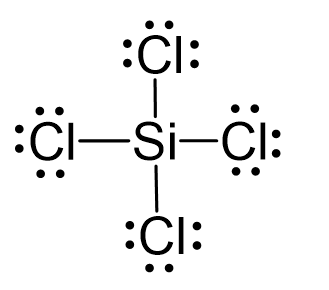
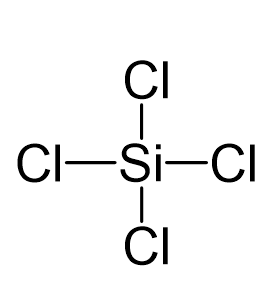
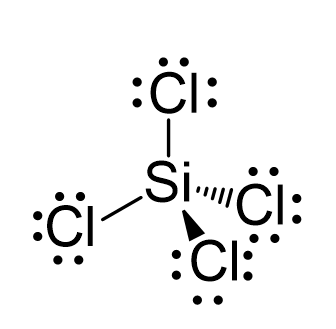
Draw the Lewis structure for SiCl4. How many bonds are around the central atom, and what is the shape of this molecule? | Homework.Study.com
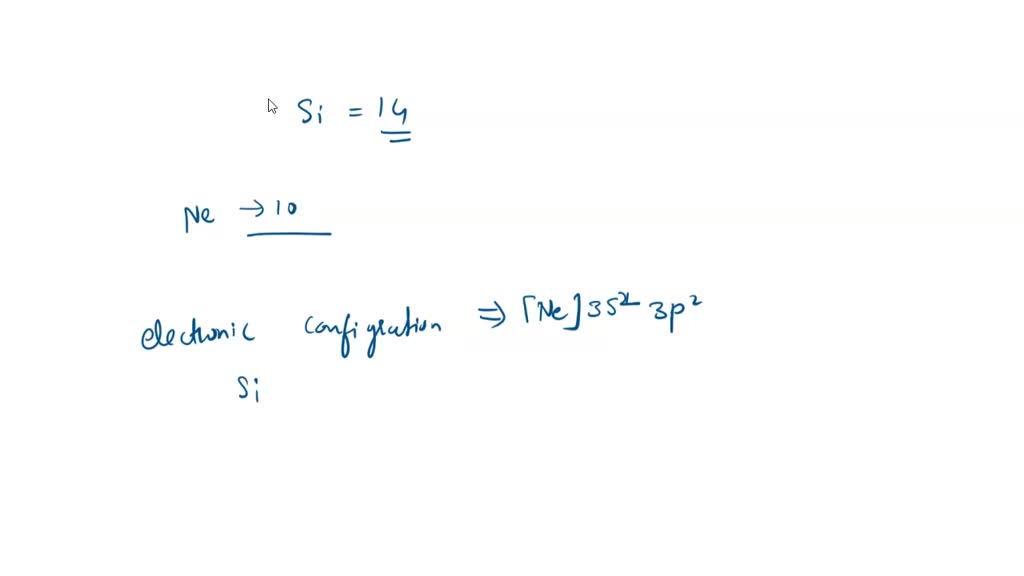
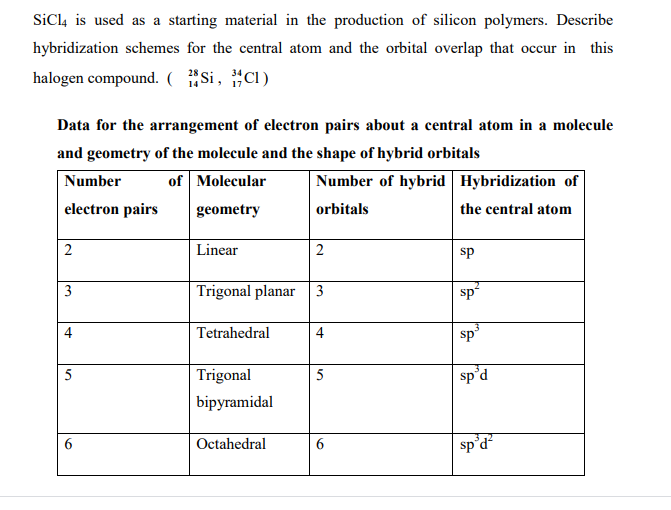
SOLVED: For the tetrahedral molecule, silicon tetrachloride, SiCl4, write the condensed electron configuration of a lone silicon atom, predict the hybridization in the molecule, and write an electron configuration for the hybridized
We all know that CCl4 and SiCl4 both compound remains in Tetrahedral structure , with a hybridization of sp³ in the central atom. Now , Si is a much larger atom than